Senators have been busy in Washington this week crafting drug pricing reform packages and trying to work out a compromise with President Trump and his administration on various bills related to healthcare reform and drug pricing. New research in oncology sees FDA approval for a breakthrough treatment and the use of organoids in liver cancer research. This and more in this week’s Innovation Partners BioBlog.
A new Senate health package includes surprisingly aggressive drug pricing reforms
Senators Lamar Alexander and Patty Murray have come out with an unexpectedly aggressive drug pricing reform plan within their suggested health package. The two senators lead the Senate health committee. The draft legislation includes several aggressive proposals that have the potential to upend the controversial business model of drug industry middlemen known as pharmacy benefit managers. Also included are moves aimed at reducing or eliminating surprise health costs and more.
READ MORE
Many oncologists will lose money under CMS’ two-sided risk payment model, study finds
A new analysis from Avalere Health predicts that more than half of all oncology practices participating in the Centers for Medicare & Medicaid Services’ (CMS) Oncology Care Model could end up owing the government money if they are required to join in a two-sided risk payment model. Beginning in July, CMS plans to require practices that have not yet achieved a performance-based payment in any of the first four performance periods of the program to switch to a two-sided risk arrangement or exit the program. This model may cause oncologists to lose a considerable amount of money.
READ MORE
FDA approves Novartis Piqray® – the first and only treatment specifically for patients with a PIK3CA mutation in HR+/HER2- advanced breast cancer
Novartis today announced the US Food and Drug Administration (FDA) has approved Piqray® (alpelisib, formerly BYL719) in combination with fulvestrant for the treatment of postmenopausal women, and men, with hormone receptor positive, human epidermal growth factor receptor-2 negative (HR+/HER2-), PIK3CA-mutated, advanced or metastatic breast cancer, as detected by an FDA-approved test following progression on or after an endocrine-based regimen The approval marks the first ever treatment specifically for HR+/HER2- advanced breast cancer with a PIK3CA mutation.
READ MORE
Trump-Pelosi fight threatens drug pricing talks
President Trump’s threat to cut off work with Democrats threatens several bipartisans issues. This includes initiatives from Senator Pelosi who has been working towards suggestions for lowering drug prices. Trump has cut off talks until the Democrat’s investigation of him stops. But in a potentially positive sign for the talks, a White House official said Thursday that discussions with Pelosi’s office on drug pricing are likely to move forward.
READ MORE
Trump to unveil executive order to force drug cost disclosure: WSJ
President Donald Trump is expected to sign an executive order as early as next week that would require for the first time disclosure of prices across the healthcare industry, in a move that officials hope will lower healthcare costs. The presidential directive would ask federal agencies to force price disclosures across the industry, including the negotiated rates between hospitals and insurers.
READ MORE
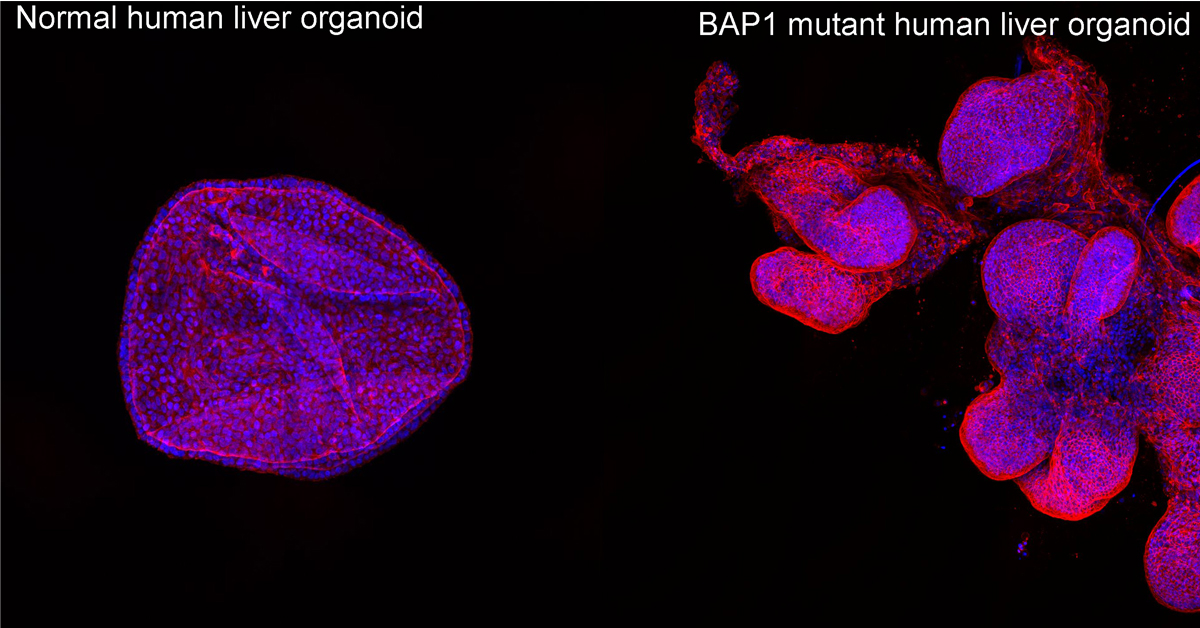
Studying the function of liver cancer genes in mini-organs
Researchers from the Hubrecht Institute and Radboud University published results from a new study in the scientific journal Cell Stem Cell. They have developed a human model in which they use organoids, or mini organs, to study the function of specific genes that are mutated in liver cancer. Using this method, they have found that mutations in BAP1, a gene commonly mutated in liver cancer, changes the behaviour of the organoids, which may make them more likely to be invasive. Organoids are mini organs that can be grown in the lab, from a very small piece of tissue derived from a variety of organs.
READ MORE




